Background:
Organisms are in a constant struggle for survival in nature. Many organisms also have the added challenge of having to face countless viral and bacterial infections. Luckily, many plants have antimicrobial properties to help defend against these diseases.
Purpose:
To discover the antimicrobial properties of various plants.
Materials:
- LB broth base
- Medium bottles, 250 mL
- Sterilizer/ autoclaves
- Water bath, 37*C, shaking
- Sterile LB agar
- Laminar flow hood and disinfectant
- Plastic safety glasses
- Bunsen burner and gas lighter
- Inoculating loop, Ni/Cr wire
- Petri dishes, 60x15mm, sterile
- E. coli JM109 (stock plate)
- Plant specimen
- Mortar and pestle
- Pipet, 10 mL and pump
- Plastic funnels, short-stemmed
- Filter paper disks, 5mm diameter
- 100 mL beakers
- Syringe, 10 mL and filter, 0.2 micrometers
- Reaction tubes and rack, 1.7 mL
- Methanol, absolute
- Pipet, 1 mL and pump
- Dry block heater/heat block
- Forceps, fine-tipped
- Ampicillin
- Glass spreader
- Incubator oven, 37*C
Procedure:
Results:
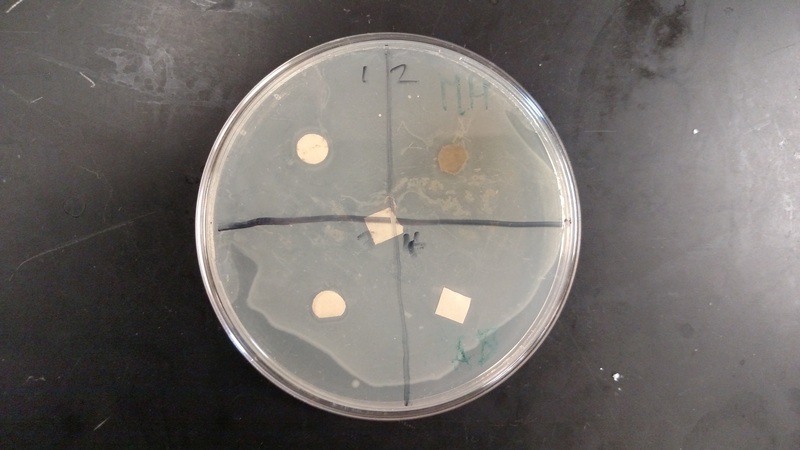
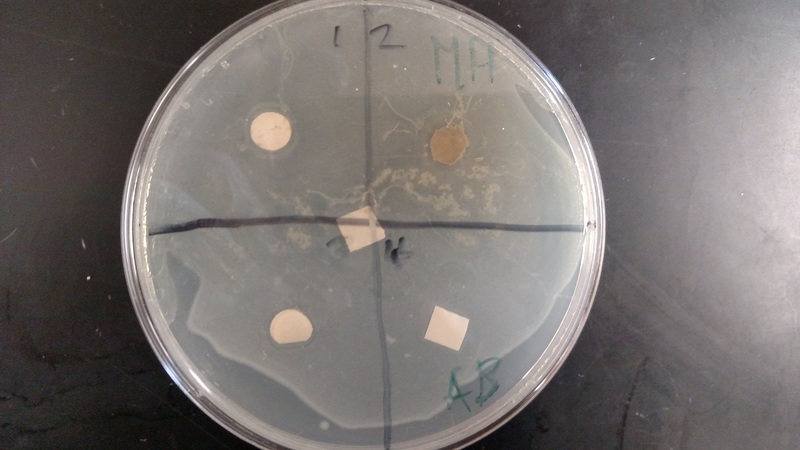
We found that the results were inconclusive due to a contamination within the sample. We concluded that any clearance areas around the samples were false clearance areas, due to the water within the samples.
Organisms are in a constant struggle for survival in nature. Many organisms also have the added challenge of having to face countless viral and bacterial infections. Luckily, many plants have antimicrobial properties to help defend against these diseases.
Purpose:
To discover the antimicrobial properties of various plants.
Materials:
- LB broth base
- Medium bottles, 250 mL
- Sterilizer/ autoclaves
- Water bath, 37*C, shaking
- Sterile LB agar
- Laminar flow hood and disinfectant
- Plastic safety glasses
- Bunsen burner and gas lighter
- Inoculating loop, Ni/Cr wire
- Petri dishes, 60x15mm, sterile
- E. coli JM109 (stock plate)
- Plant specimen
- Mortar and pestle
- Pipet, 10 mL and pump
- Plastic funnels, short-stemmed
- Filter paper disks, 5mm diameter
- 100 mL beakers
- Syringe, 10 mL and filter, 0.2 micrometers
- Reaction tubes and rack, 1.7 mL
- Methanol, absolute
- Pipet, 1 mL and pump
- Dry block heater/heat block
- Forceps, fine-tipped
- Ampicillin
- Glass spreader
- Incubator oven, 37*C
Procedure:
- Grind up 2 grams of plant tissue from leaves or bark with your mortar and pestle with 10 mL of de-ionized water. Rest for 3 minutes. Then filter the sample through the 11 cm filter paper funnel. With a syringe filter, sterilize the filtered sample extract. Collect 1 mL of the extract into a 1.7 mL microbe, and be sure to label it. Do step 4 again, but replaced the deionized water with methanol. After you extract the methanol, place the 1.7 mL tube with the extract in a 65*C heat block with caps open for 24+ hours to evaporate the methanol.
- Sterilization: Attach prefilter to syringe and rinse with water. Take to Laminar Hood (plant extract syringe/pre-filter, pipet). Label microfuge tube. (initials, W or M). Attach sterile filter to pre-filter. Load 1.7 ml of extract into syringe using pipet. Depress plunger- at least 1 ml. Snap on cap without touching inside. For the rest of your samples, do steps 4 and 5 again, making sure to label all samples. There should be 2 tubes in total.
- Evaporate methanol from methanol extract by placing tube, with cap open, on a 65 C heat block overnight.
- Reconstitute methanol extract with 1.0 ml sterile deionized water. With sterilized forceps that have been flamed in alcohol, drop 3 filter paper disks into every tube of filtered extract.
- Make the negative control disks: three each of only the methanol and only the distilled water. Make 2 positive control disks of the ampicillin solution.
- Let the disks soak up enough extract to be saturated. This may have to happen overnight.
- Sterile disks were added to microfuge tubes containing 1 mL sterile water and 1 mL ampicillin.
- 10-20 mL of warmed nutrient agar was poured into 2 petri dishes using sterile technique. Close all tubes, and store the samples at 4*C until time to use.
- With a sterile pipet, transfer 1 mL of the prepared E. coli broth to the middle of the Petri dish. Then get a glass spreader, sterilize it using alcohol and flame, and spread the broth evenly across the dish. Cover and allow the culture to soak into the agar for at least 15 minutes.
- With sterile forceps (alcohol and flame), place one disk into the middle of each quadrant (which you should have drawn in). Blot out the extra liquid on the disks before you place them on the 2 Petri dishes. Keep the methanol-extracted samples in one dish and the water in the other.
- Put one of the negative control disks (with methanol or distilled water) in the marked area. Do the same with the positive ampicillin soaked one. Incubate the petri dish at 37*C overnight upside down. Examine each quadrant and the controls for areas of inhibition. Photograph and draw the results.
Results:
We found that the results were inconclusive due to a contamination within the sample. We concluded that any clearance areas around the samples were false clearance areas, due to the water within the samples.